
Earth’s Water – Fallen from Outer Space or Degassing from Inside the Planet?
This is certainly a big question. That is, did nearly all of Earth’s water arrive from outer space or has it always been present in the Earth’s mantle and released to the surface over time? We will not be answering it definitively here, of course, but knowledgeable scientists continue to debate the alternatives. It is not a closed subject. We will review a few of the salient points scientifically asserted because they relate closely to the idea of Deep Seated Water, a vast proportion of which may originate from deep in the Earth.
Where Did the Abundant Water on Our Planet Come From?
Given the number of barren rocks in the solar system and beyond, a good question to ask is “Where did the abundant water on our planet come from?” There is a lot of frozen water in the outer solar system and moons made of it orbiting the large gaseous planets. The sun’s gravity pulls comets well down into the inner solar system in great elliptical orbits, falling, it’s said, from the Oort Belt of presumed icy objects far out on the fringe of our system. It is commonly taught in school that early in the Earth’s history, these great comets repeatedly struck the Earth bringing water to our oceans and, in fact, providing virtually all the water on Earth. The idea is, we’re drinking and swimming in comet water.

But that has always been only a hypothesis. What backs the case? Certain asteroids in our solar system known as carbonaceous chondrite have been identified carrying not just heavy metals, but also ‘volatile’ substances like carbon and water, which are necessary for essentially all life forms on earth.[1]1 So water has been found on asteroids. But does this account for all of the water in the oceans?
Do Comets Really Account for all of the Water in the Oceans?
Many recent studies are poking holes in that scientific conjecture. For example, one study from January 2017 revealed that one iron-loving heavy metal in the Earth’s mantle has been found to have a different atomic signature than the metal on asteroids commonly found in the outer solar system (where all those ice moons are).[2] 2 Volatile substances like water are in fact rare in the inner solar system, the inference being that the “late veneer”[3]3 of our planet was formed by matter from closer in to the sun (the volume without water.) A pretty technical point, admittedly, but that is the science.
Another point is that comets carrying large quantities of volatiles, even if they did impact early Earth, would most likely not have carried their load of water, etc., all the way to the surface. A study of a great impact crater in Canada[4]4 revealed that most of the element ‘lead’ had been completely volatized from it before impact. Certainly, the even more volatile substance of water would also have been lost to space as well. It’s even asserted that the planet may lose mass from some of these early great impacts.
Lastly, geochemists[5]5 have asserted that the Earth’s oceans contain different amounts of ‘heavy’ hydrogen than celestial asteroids and comets. It’s not the same water. We could go on. The fact is that the comet theory is getting increasingly shot full of holes.
What is the Alternative Theory of the Source of the Earth’s Water?
What is the alternative theory of the source of water: that it has always been present in various rocks of the Earth’s mantle and that this water has been released to the surface continuously over time (up to the present day)…through volcanoes.
Balz Kamber, Chair of Geology and Mineralogy at Trinity College Dublin, states the case rather succinctly:
“…evidence suggests that the Earth’s surface water or ‘liquid hydrosphere’ actually accumulated by degassing from within the planet. Water is stored in the Earth’s mantle in the form of hydroxyl groups (one hydrogen and one oxygen atom) trapped within minerals such as ringwoodite.
When the mantle rocks melt, the water dissolves into the magma. As the magma rises towards the surface and cools, pressure is reduced, crystals form and the water is released and emitted as vapour through volcanoes. With this mechanism, water from great depth can be degassed to the surface.”[6]6
What are the facts supporting this? They are many. Here are a few: The first is that complex studies show that the water on our planet is as old as it is, at least, 3.85 billion years. [7]7That is, it did not arrive after the mass of the planet was formed but was here from the beginning. Other unique studies have found, as well, that minerals at depths up to 200 kilometers do contain water.[8]8 More recent seismic and modelling data increases that depth to almost 700 kilometers below the surface. [9]9Some of the same scientists, studying how diamond is formed (at great depth), suggest that water could be found even deeper.[10]10
Water might be deep down there, melded with rock, but there is a lot of water up here in our oceans and lakes. How does that buried water get to the surface?
Volcanoes’ Role
The U. S. Geological Survey in an essay[11]11 on the composition of volcanic gases states:
“Magma contains dissolved gases, which provide the driving force that causes most volcanic eruptions. As magma rises towards the surface and pressure decreases, gases are released from the liquid portion of the magma (melt) and continue to travel upward and are eventually released into the atmosphere.
Large eruptions can release enormous amounts of gas in a short time. The 1991 eruption of Mt. Pinatubo is thought to have injected more than 250 megatons of gas into the upper atmosphere on a single day. However, even if magma never reaches the surface, gases can often escape continuously into the atmosphere from the soil, volcanic vents, fumaroles, and hydrothermal systems.
“By far the most abundant volcanic gas is water vapor…”
The last sentence is, of course, key to our discussion. Multiple scientific references assert that water has composed between 90% and 97% in measurements of the composition of volcanic gases.[12]12

Mount Nyiragongo Volcanic Eruption Causes Panic. Photo by Pierre-Yves Burgi on Unsplash.
Volcanoes, of course, are not distant history but impact our world in the present time. Case in point: Mount Nyiragongo erupting on May 22, 2021, perilously close to Goma, Democratic Republic of Congo. It might bring water vapor, but also wreaks havoc on nearby populations. The very reason that water vapor is being introduced constantly to our atmosphere is because the Earth’s crust is alive.
 Another modern volcano: Mt. St. Helens, exploding only 40 miles outside Portland, Oregon on May 18, 1980.
Another modern volcano: Mt. St. Helens, exploding only 40 miles outside Portland, Oregon on May 18, 1980.
It is the largest and most explosive volcanic eruption that release the most gaseous water vapor. This is apparently because they source from deep in the magma which contains dissolved water under great pressure. The lessening of the pressure releases the superheated water vapor, which expands 1000 times when it transforms from its compressed, dissolved phase in magma into its gaseous phase, and that is what creates the eruptive force that impels the magma upward to find explosive release.
The consistency of the magma that is sourcing the volcano changes its gaseous atmospheric release. The volcanoes in Hawaii, for example, release gases rich in sulfur and carbon compounds but contain less water, which explains why such island volcanoes far from continental margins are not very explosive – less water in that source magma. On the other hand, the most famous, highly explosive volcanoes, including especially the ‘Ring of Fire’ surrounding the edges of the Pacific Basin, release magma of a consistency that contains much more water. [13]13
Long before the explosion of Mount Pinatubo in the Philippines (referenced above), there was, of course, Krakatoa in Sumatra which in 1883 utterly vaporized an island in an area where there had been a history of similar colossal eruptions. Computations estimate it to have released 6 cubic miles of rock. Based on what we know now, how much of that (or more) was water vapor? An earlier super-volcano, Tambora of an 1815 eruption, has been estimated to have released one billion cubic kilometers of magmatic water vapor into the stratosphere.[14]14 With sixty to eighty volcanic eruptions a year of various sizes and gaseous compositions, the Earth’s atmosphere and hydrologic cycle are receiving a constant and significant quantity of new water. And this has been going on from, so to speak, Day One.
So, whether or not early on cometary impacts contributed partially to the Earth surface’s store of water, there is overwhelming evidence that the Earth’s magma contributes massively on a regular basis.
And water does not come up only in these great, highly observable explosions. There are volcanic submarine explosions such as this recent one between Fiji, Tonga, and Samoa.

Submarine volcanic eruption. Photography courtesy of NOAA and NSF.
Add in continuous diffuse geothermal seeps of (often very hot) water from the sea floor, such as this in the Sea of Cortez outside Mexico.

Sea of Cortez outside Mexico. Photograph by Brian J. Skerry, National Geographic
Imagine how many such seeps Man knows nothing about, distributed across the vast floor of the oceans. Then there are the famous recently discovered extremely hot, perennial hydrothermal vents (‘white’ and ‘black’) from the sea floor, including as deep as the Marianas Trench, which are distributed principally along the tectonic boundaries under continuous crustal pressures, as in this map.
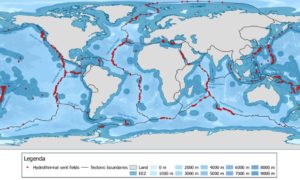
Hydrothermal Vents. Map created making use of the InterRidge ver.3.3 database.
There are hydrothermal vents not associated with the tectonic plate intersections, such as those coming up from deep within the caldera created from an earlier mega-explosion that destroyed one island, creating three in its wake, the Maug Islands on the northern rim of the Marianas.
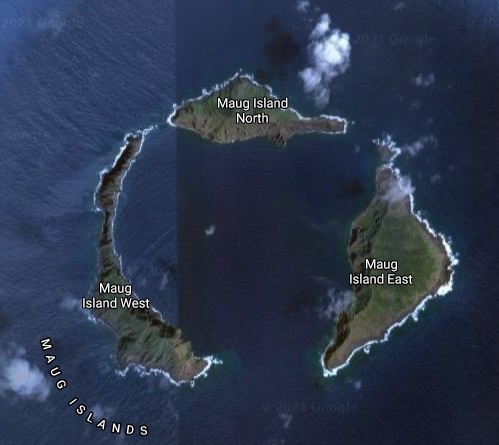
Maug Islands on the northern rim of the Marianas
The list goes on and on. How much water have all of these sources cumulatively added to the oceans and the Earth’s surface across millions, if not billions of years? How much are they adding to this day? It is not an inconsequential figure.
An Enormous Store of Water
In summation, there is abundant, if not overwhelming evidence, that there is an enormous store of water in the magmas underlying the Earth’s crust and that these are being released through multiple avenues to the surface, sometimes in greater and sometimes lesser volumes, as illustrated in part in this graphic created by the U.S. Geological Survey:
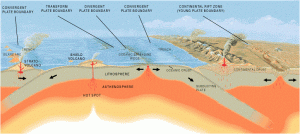
Illustration by José F. Vigil from “This Dynamic Planet” – A wall map produced jointly by the U.S. Geological Survey, the Smithsonian Institution, and the U.S. Naval Research Laboratory.
This shows that ocean vents are found near mid-ocean divergent plate boundaries (tectonic plates being ripped apart) as well as in volcanic arcs associated with convergent tectonic plate boundaries (plates being smashed together, causing subduction.)
There are, of course, volcanoes and geothermal vents not at sea, but on the major continents and larger islands (such as Iceland, for example). Where magma has not been forced up to break the surface, it of course can make its way up through cracks, fractures, weaker rock formations, etc. to approach the surface, where aquifers of water, more or less deep, may store between aquitards (barrier formations) for long periods of time and be accessible where techniques are known how to locate them. Some of these magmatic-sourced aquifers may be being continuously replenished from deep-reaching fractures.
This brings us back to Deep Seated Water and how to find it. The short, informal essay above provides a general background affirming that there is more than ample evidence of an enormous resource of water much deeper than the surface hydrological cycle, water infused chemically in and with magma. The subject of how to discover and access stores or aquifers of such water that have been forced up close to the Earth’s surface is for future essays.
Clark Carr
Director of Field Operations
Lawrence Anthony Earth Organization
Updated 29 March 2023, 8 October 2021
1 June, 2021
Footnotes
[1] Wing, M.R., Bada, J.L. The origin of the polycyclic aromatic hydrocarbons in meteorites. Origins Life Evol Biosphere 21, 375–383 (1991). https://doi.org/10.1007/BF01808308
[2] Fischer-Gödde, M., Kleine, T. Ruthenium isotopic evidence for an inner Solar System origin of the late veneer. Nature 541, 525–527 (2017). https://doi.org/10.1038/nature21045
[3] “Late Veneer”: The term late veneer refers to the late accretion of asteroidal or cometary material to terrestrial planets. Iron and nickel segregation during core formation leaves the mantle of the planets depleted in siderophile elements, notably platinum-group elements. The modern abundances of these elements in the terrestrial mantle greatly exceed the level expected from such a wholesale removal of metal. It is therefore surmised that 0.5–1.5% of chondritic or cometary material was brought to the planet by the late veneer after core formation (Dauphas and Marty 2002; Maier et al. 2009). This hypothesis is germane to the issue of water delivery to the Earth. (Definition from the Encylopedia of Astrobiology) https://link.springer.com/referenceworkentry/10.1007%2F978-3-642-11274-4_870
[4] O’Sullivan E.M., Goodhue R., et al, Chemistratigraphy of the Sudbury Impact basin fill: Volatile metal loss and post-impact evolution of a submarine impact basin, Geochimica et Cosmochimica Acta, 183, 198-233 (2016) https://doi.org/10.1016/j.gca.2016.04.007
[5] Blake, G., Qi, C., Hogerheijde, M. et al. Sublimation from icy jets as a probe of the interstellar volatile content of comets. Nature 398, 213–216 (1999). https://doi.org/10.1038/18372
[6] Kamber, B., “Comets or volcanoes? Scientists are changing their minds about how the Earth’s water got here,” The Conversation, Mar 6, 2017
[7] Nutman, A.P., Mojsis, S.J., et al, Recognition of ≥3850 Ma water-lain sediments in West Greenland and their significance for the early Archaean Earth, Geochimica et Cosmochimica Acta, 61 Iss 12, (2476 – 2484), 1997.
[8] Ulmer P., Tromsdorff V., Serpentine Staability to Mantle Depths and Sub-duction-Related Magmatism, Science, 268 Iss 5212 (1995) https://doi.org/10.1126/science.268.5212.858
[9] Schmandt B., Jacobson S.D., et al, Dehydration melting at the top of the lower mantle, Science, 344 Iss 6189, (1265-1268), 2014
[10] Palot M., Jacobson S.D, et al, Evidence for H20-bearing fluids in the lower mantle from diamond inclusion, Lithos, 265, (237-2430, 2016
[11] https://www.usgs.gov/natural-hazards/volcano-hazards/volcanic-gases
[12] Gerlach, T.M., Volcanic Gases. In: Encyclopedia of Geochemistry, Springer, (656 – 657)
And
Delmelle, P., Stix, J., Volcanic Gases. In: Encyclopedia of Volcanoes, Academic Press, (803 – 815), 1999
[13] https://www.sandatlas.org/volcanic-gases/
[14] Glaze, L.S., Baloga, S.M., Transport of atmospheric water vapor by volcanic eruption columns, Journal of Geophysical Research, Vol 102, (6099 – 6108), 1997.